4-hydroxy-N,N,N ( 4-HO-TMT) Guide: Effects, Benefits and Legal Status
4-hydroxy-N,N,N ( 4-HO-TMT) Guide: Effects, Benefits and Legal Status
- What Is 4-HO-TMT?
- How does 4-HO-TMT work?
- Natural Sources of 4-HO-TMT
- Synthetic Sources of 4-hydroxy-N,N,N
- 4-HO-TMT Chemical Structure
- 4-HO-TMT Mechanism of Action
- 4-HO-TMT Effects
- 4-HO-TMT Cognitive Effects
- 4-HO-TMT Physical Effects
- 4-HO-TMT Risks
- 4-HO-TMT Legality
- Bottom Line
What Is 4-HO-TMT?
4-Hydroxy- N, N, N-trimethyltryptamine, or 4-HO-TMT, is a chemical that forms after the body metabolizes aeruginascin, a naturally occurring compound found in certain psilocybin-containing mushrooms. 4-HO-TMT shows the potential to be psychoactive, possibly affecting mood and even inducing psychedelic experiences. Scientists theorize that 4-HO-TMT’s effects could relate to its ability to bind with specific serotonin receptors. Other theories implicate 4-HO-TMT in the entourage effect, amplifying the properties of mushrooms’ other psychedelic compounds.
Let’s explore.
How does 4-HO-TMT work?
4-HO-TMT (4-Hydroxy- N, N, N-trimethyltryptamine) is the metabolite of aeruginascin that binds to 5HT2A serotonin receptors, potentially triggering psychedelic effects. There is significant interest in 4-HO-TMT and aeruginascin following one researcher's hypothesis that the compounds could increase the chances of having a “good trip.”
Natural Sources of 4-HO-TMT
Researchers don't know if magic mushrooms naturally produce 4-HO-TMT. To date, they only know that the compound is a metabolite of aeruginascin, which occurs naturally in specific species of psilocybin mushrooms, like Inocybe aeruginascens, Pholiotina cyanopus, and Psilocybe cubensis.
Synthetic Sources of 4-hydroxy-N,N,N
4-HO-TMT may exist in nature, but scientists are studying its synthetic version in the lab. In 2020, Andrew Chadeayne, founder of the pharmaceutical drug discovery company CaaMTech, led a joint venture with Canopy Growth USA, successfully synthesizing 4-HO-TMT. The results motivated CaaMTech to develop a proprietary analog called Amphora, which has a similar structure and will undergo further study.
4-HO-TMT Chemical Structure
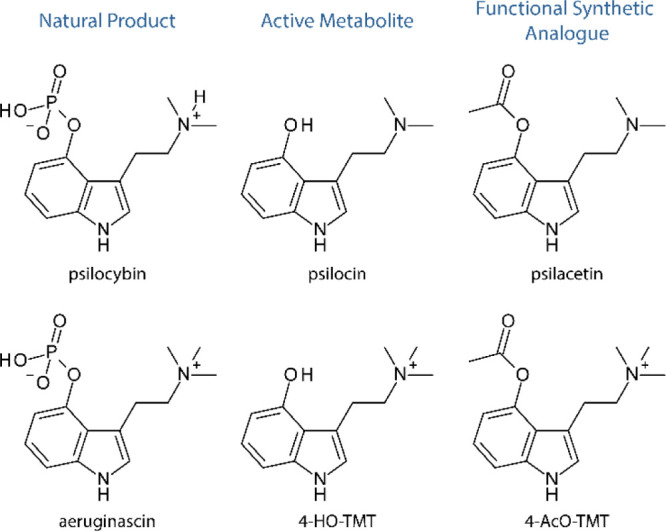
4-HO-TMT is a tryptamine alkaloid (psychoactive compound) with two methyl groups attached to its tryptamine core. Its molecular structure is almost identical to aeruginascin, except it lacks a phosphate group lost during metabolization.
4-HO-TMT's molecular structure makes it part of the quaternary ammonium group (QAG.) This classification has stirred up debate regarding its ability to cross the blood-brain barrier and affect brain function. Quaternary ammoniums are compounds with large size, positive charge, and poor fat solubility, generally believed not to cross the blood-brain barrier easily. However, researchers investigating 4-HO-TMT have made a case for possible exceptions, noting a record of some quaternary ammoniums entering the brain.
Need a Test or Have a Question
4-HO-TMT Mechanism of Action
Studies on 4-HO-TMT show that it has a strong affinity with the 5HT2A receptor associated with learning, memory, neurogenesis, desensitization, and re-sensitization. 5HT2A, found in the brain, gut, and cardiovascular system, is the same receptor that psilocybin, LSD, and DMT bind with to create psychedelic effects.
4-HO-TMT also effectively binds to 5HT1A and 5HT2B receptors, influencing and regulating several bodily functions, including mood and physiological processes.
- 5HT1A some text
- Increases dopamine release in the medial prefrontal cortex, striatum, and hippocampus.
- Induces peripheral vasodilation and stimulates the vagus nerve.
- 5HT2Bsome text
- Inhibits serotonin and dopamine uptake.
- Regulates cardiac structure and functions.
4-HO-TMT Effects
Currently, no published testing of 4-HO-TMT in humans or animals exists. Its robust interaction with the 5HT2A receptor shows psychedelic promise, but whether it can produce full mind-altering effects depends on its ability to cross the blood-brain barrier.
4-HO-TMT Cognitive Effects
Assuming 4-HO-TMT binds to 5HT2A receptors in the brain, it would likely induce psychedelic effects, affecting mood, perception, and potentially hallucinations. However, further research is necessary to confirm this. Other research suggests it could elevate mood without inducing hallucinations.
The chemist, Jochen Gartz, who discovered aeruginascin, proposed that 4-HO-TMT and aeruginascin could increase the chances of a good trip. He made these conclusions based on lab analysis of reports from 23 individuals who consumed aeruginascin-containing mushrooms and reported only positive experiences. The mechanism behind this effect is unknown, but CaamTech developed a 4-HO-TMT compound and is sponsoring research based on this theory.
4-HO-TMT Physical Effects
An online theory suggests that aeruginascin, and by association, 4-HO-TMT, could be partially responsible for a phenomenon known as "wood lover paralysis," which causes temporary paralysis in some individuals.
An early theory by Andrew Chadeayne suggests 4-HO-TMT and aeruginascin could be catalysts for “wood lovers' paralysis” due to their similarity to bufotenine, a compound found in psychedelic toad venom from "Bufo" toads. Bufotenidine binds to a receptor known as 5HT3A, which can interfere with the nerves that mediate muscle contractions.
However, a later review of 4-HO-TMT's interactions with various serotonin receptors indicates that it does not interact with the 5HT3A receptor. If 4-HO-TMT is responsible for paralysis, it will act through a different mechanism.
4-HO-TMT Benefits
Researchers think 4-HO-TMT may contribute to positive trips and magic mushroom benefits through the "Entourage Effect". Coined by the cannabis industry, the Entourage Effect refers to the idea that plant compounds synergize in the body to amplify the overall therapeutic and psychoactive outcomes. However, further research is needed to explore the pharmacology of other naturally occurring tryptamine compounds.
4-HO-TMT Risks
As a general rule, tryptamines like 4-HO-TMT have a good safety profile. However, biochemistry is complex, and until scientists conduct clinical trials, no one can make assertions regarding potential risks.
Some literature expresses concerns about tryptamines interacting with serotonin and 5HT2B receptors after a study showing a connection to cardiac valvulopathy (heart valve disease). Still, researchers mention 4-HO-TMT's low binding affinity to 5HT2B, suggesting cardiac concerns may not be an issue.
4-HO-TMT Legality
4-HO-TMT is not an illicit scheduled substance anywhere in the world. However, due to its similar molecular structure to psilocybin, some laws might consider it an analog of psilocybin. The Federal Analogue Act in the United States could cause legal troubles for individuals selling or transporting 4-HO-TMT. However, no legal proceedings around 4-HO-TMT are on record.
Bottom Line
4-HO-TMT, a metabolite of aeruginascin found in certain species of psilocybin mushrooms, shows potential psychoactive effects and may contribute to mood modulation and psychedelic experiences. Its mechanism of action involves binding to serotonin receptors, particularly the 5HT2A receptor. However, human studies on 4-HO-TMT are lacking, and its ability to cross the blood-brain barrier and induce mind-altering effects is still uncertain. The compound's physical impact, benefits, and risks require further investigation, particularly its role in the entourage effect and potential cardiac concerns. Legally, 4-HO-TMT is not classified as a scheduled substance, but its analog status could lead to illicit implications in some jurisdictions. Continued research is necessary to understand the properties and impact of 4-HO-TMT fully.
ACS Laboratory tests 4 HO TMT along with 7 other tryptamines in psychedelic mushrooms from DEA-registered facilities. Below is an example of a Certificate of Analysis published with permission from our client. Contact us today to start testing.




.jpg)




.png)
.png)
.png)
.png)
.png)